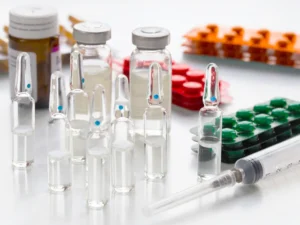
Pharmaceutical companies worldwide are under increasing pressure to design safer, smarter and more sustainable packaging. Regulatory bodies, environmental regulations and patient safety requirements are reshaping how packaging materials are selected. A comprehensive Pharma Packaging material selection guide – biocompatible, sustainable and recyclable in 2026 requirement helps pharmaceutical manufacturers ensure compliance, sustainability and product protection.
In modern pharmaceutical development, packaging is no longer just a container. It is a critical engineering system that protects drugs, maintains stability, improves patient safety and supports sustainable supply chains.
Mechanical product design and development companies such as Nirvipa Technology play a key role in developing next-generation pharmaceutical packaging solutions by combining material science, product design, simulation and manufacturing expertise.
This guide explains how to select pharmaceutical packaging materials that meet biocompatibility, sustainability, recyclability and regulatory compliance requirements for 2026 and beyond.
Why pharmaceutical packaging material selection matters in 2026

The pharmaceutical industry is rapidly evolving due to stricter regulations, global supply chains and sustainability commitments. Packaging plays a major role in maintaining drug safety and effectiveness throughout its lifecycle.
Poor packaging material selection can cause several problems:
-
Drug contamination
-
Chemical reactions between drug and packaging
-
Moisture or oxygen exposure
-
Reduced shelf life
-
Regulatory non-compliance
Modern pharmaceutical packaging must protect products from environmental conditions such as light, moisture, oxygen, temperature fluctuations and microbial contamination.
A well-structured Pharma Packaging material selection guide – biocompatible, sustainable and recyclable in 2026 requirement helps companies evaluate materials using scientific testing and engineering analysis.
Engineering teams often conduct:
-
-
Material compatibility studies
-
Mechanical strength testing
-
Sterilization validation
-
Supply chain durability analysis
-
This ensures pharmaceutical products reach patients safely and reliably.
Best biocompatible materials for pharmaceutical packaging

Biocompatibility is one of the most critical factors when selecting pharmaceutical packaging materials. Packaging materials must not interact chemically with the drug formulation or cause harmful reactions.
Several materials are widely used due to their chemical stability and regulatory acceptance.
1. Medical-Grade polymers
Medical-grade polymers are commonly used for pharmaceutical packaging because they are lightweight, durable, and compatible with sterilization processes.
Common polymers include:
-
Polypropylene (PP)
-
High Density Polyethylene (HDPE)
-
Cyclic Olefin Polymer (COP)
-
Cyclic Olefin Copolymer (COC)
These polymers are often used in:
-
Syringes
-
Drug cartridges
-
Inhaler devices
-
Injectable drug containers
They provide excellent chemical resistance, transparency, and dimensional stability.
2. Pharmaceutical glass
Glass has long been considered the gold standard for pharmaceutical packaging because it is chemically inert.
Types of pharmaceutical glass include:
-
Type I Borosilicate Glass
-
Type II Treated Soda-Lime Glass
-
Type III Soda-Lime Glass
Advantages of glass packaging include:
-
Excellent chemical resistance
-
High transparency
-
Strong barrier properties
However, glass is heavier and more fragile compared to polymer packaging.
3. Advanced barrier polymers
Advanced materials such as multi-layer polymer composites and nano-barrier coatings are gaining popularity. These materials provide improved protection against oxygen and moisture while maintaining lightweight structures.
Mechanical engineering teams often use advanced simulation tools to validate material compatibility and structural performance.
Biocompatible medical packaging materials and testing

Biocompatibility testing ensures that packaging materials do not release harmful substances into drug formulations.
International regulatory standards include:
-
ISO 10993 biological evaluation
-
USP <87> cytotoxicity testing
-
USP <88> biological reactivity testing
Testing procedures evaluate:
-
Extractables and leachables
-
Toxicity levels
-
Material degradation
Engineering teams working on pharmaceutical packaging development must integrate material science research with design validation.
This is where product design companies like Nirvipa Technology contribute by performing:
-
Material compatibility studies
-
Prototype testing
-
Engineering validation
These processes ensure packaging meets strict regulatory requirements.
Sustainable pharmaceutical packaging materials

Sustainability is becoming a top priority for pharmaceutical manufacturers worldwide. Governments and regulatory bodies are introducing policies aimed at reducing plastic waste and carbon emissions.
Eco-friendly pharmaceutical packaging materials help companies meet these environmental goals.
Recyclable plastics
Common recyclable materials include:
-
PET (Polyethylene Terephthalate)
-
HDPE
-
Polypropylene (PP)
These materials are widely accepted in recycling systems and are frequently used in pharmaceutical bottles and containers.
Bio-based polymers
Bio-based polymers are derived from renewable sources such as corn or sugarcane.
Examples include:
-
Polylactic Acid (PLA)
-
Bio-Polyethylene
-
Polyhydroxyalkanoates (PHA)
These materials reduce dependence on fossil fuels and support circular economy initiatives.
Paper-based secondary packaging
Paperboard packaging is widely used for:
-
Medicine cartons
-
Instruction leaflets
-
Blister pack backing cards
Many pharmaceutical companies now prefer FSC-certified paperboard materials.
Recyclable packaging design strategies
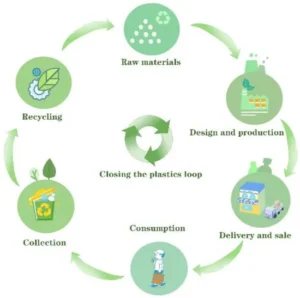
Recyclability is influenced not only by the material but also by the packaging design. Engineering teams must develop packaging structures that allow efficient recycling.
Mono-material packaging
Mono-material packaging uses a single polymer rather than multi-layer materials. This simplifies recycling processes and improves sustainability performance.
Reduced material usage
Lightweight packaging reduces environmental impact by lowering raw material consumption and transportation emissions.
Design for disassembly
Packaging should allow easy separation of components such as caps, seals, and labels.
Mechanical engineers often implement design for recyclability principles during the packaging development phase.
Role of mechanical product design in pharma packaging development
Mechanical engineering plays a crucial role in pharmaceutical packaging innovation. Design teams perform multiple engineering activities to ensure packaging reliability and manufacturability.
Key services include:
-
3D CAD packaging design
-
Finite Element Analysis (FEA)
-
Injection molded packaging development
-
Rapid prototyping
-
Structural performance testing
Engineering companies such as Nirvipa Technology support pharmaceutical manufacturers with product design and development services that accelerate packaging innovation.
These services help companies:
-
Reduce development time
-
Improve packaging safety
-
Optimize manufacturing processes
-
Ensure regulatory compliance
By integrating engineering expertise with material science, companies can develop next-generation pharmaceutical packaging solutions.
Engineering challenges in pharma packaging material selection
Selecting the right pharmaceutical packaging materials is not only a regulatory requirement but also a complex engineering challenge. Modern drug formulations, especially biologics, vaccines and combination products, require packaging systems that maintain stability, sterility and safety throughout the product lifecycle. This makes the Pharma Packaging material selection guide – biocompatible, sustainable and recyclable in 2026 requirement increasingly important for pharmaceutical manufacturers and packaging developers.
One of the primary challenges is material compatibility with advanced drug formulations. Many modern biologic drugs are highly sensitive to environmental factors such as oxygen, moisture, and light exposure. Packaging engineers must carefully analyze the barrier properties of materials to ensure the drug remains stable during storage and transportation. This often requires advanced testing methods such as extractables and leachables studies, accelerated aging tests, and environmental simulations.
Another challenge involves balancing sustainability goals with regulatory compliance. Pharmaceutical companies are under pressure to reduce plastic waste and adopt recyclable packaging solutions. However, sustainable materials must still meet strict regulatory standards for drug safety and patient protection. Engineers must therefore design packaging systems that maintain high performance while minimizing environmental impact.
Manufacturing feasibility is also a key factor in packaging material selection. Materials must be compatible with large-scale production processes such as injection molding, thermoforming, and automated assembly lines. Design teams must optimize packaging structures to ensure efficient manufacturing, cost-effectiveness, and supply chain reliability.
Engineering service providers like Nirvipa Technology help pharmaceutical companies address these challenges by offering specialized mechanical product design and development services. Through advanced CAD modeling, rapid prototyping, and design validation testing, engineering teams can develop innovative pharmaceutical packaging solutions that meet the evolving requirements of biocompatibility, sustainability, and recyclability in 2026 and beyond.
By combining engineering expertise with material science and regulatory knowledge, pharmaceutical companies can create packaging systems that protect drugs, support sustainability goals, and enhance patient safety across global markets.
Emerging trends in pharmaceutical packaging for 2026
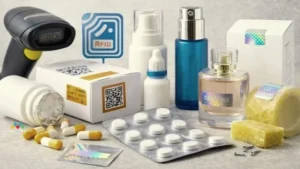
The pharmaceutical packaging industry is experiencing rapid innovation driven by digital technology and sustainability initiatives.
Smart packaging
Smart packaging systems include sensors and digital identifiers that monitor:
-
Temperature
-
Humidity
-
Drug authenticity
-
Supply chain tracking
Anti-counterfeiting technologies
To combat counterfeit drugs, pharmaceutical packaging now incorporates:
-
QR codes
-
NFC tags
-
Tamper-evident seals
Connected healthcare packaging
Connected packaging integrates digital health platforms to monitor patient medication adherence.
AI-Driven packaging design
Artificial intelligence tools help engineers optimize packaging materials, structural designs, and sustainability performance.
These innovations will continue shaping pharmaceutical packaging beyond 2026.
Frequently asked questions (FAQs)
1. What is the most important factor in pharmaceutical packaging material selection?
Drug compatibility is the most important factor because packaging materials must not react with the drug formulation.
2. What materials are commonly used in pharmaceutical packaging?
Common materials include glass, polypropylene, HDPE, cyclic olefin polymers, aluminum foil laminates, and medical-grade plastics.
3. Why is sustainable pharmaceutical packaging important?
Sustainable packaging reduces environmental impact, meets regulatory requirements, and supports global circular economy initiatives.
4. What is biocompatibility in pharmaceutical packaging?
Biocompatibility ensures packaging materials do not cause harmful reactions when in contact with drugs or patients.
5. What are recyclable pharmaceutical packaging materials?
PET, HDPE, PP, and certain paperboard materials are widely recyclable in global recycling systems.
6. How does mechanical product design support pharma packaging development?
Mechanical engineering helps design packaging structures, validate materials, optimize manufacturing, and ensure regulatory compliance.
Conclusion
Selecting the right packaging materials is critical for pharmaceutical product safety, regulatory compliance, and environmental sustainability. A well-structured Pharma packaging material selection guide – biocompatible, sustainable and recyclable in 2026 requirement helps pharmaceutical companies develop packaging systems that protect drugs and meet modern regulatory expectations.
Biocompatible materials ensure patient safety, while sustainable and recyclable packaging supports environmental responsibility. Mechanical product design and development services further enable companies to create innovative packaging solutions that are reliable, scalable, and compliant.
By partnering with engineering experts like Nirvipa Technology, pharmaceutical companies can accelerate product development and build future-ready packaging systems that meet the evolving demands of the healthcare industry.
👉 Explore our services and discover how we can help you for your new Pharma packaging design and development innovation solution.
🔗 Visit: https://www.nirvipa.com
